













| Production Scores: | Multiple Varieties |
|---|---|
| Production Continuity: | Continuous |
| Condition: | New |
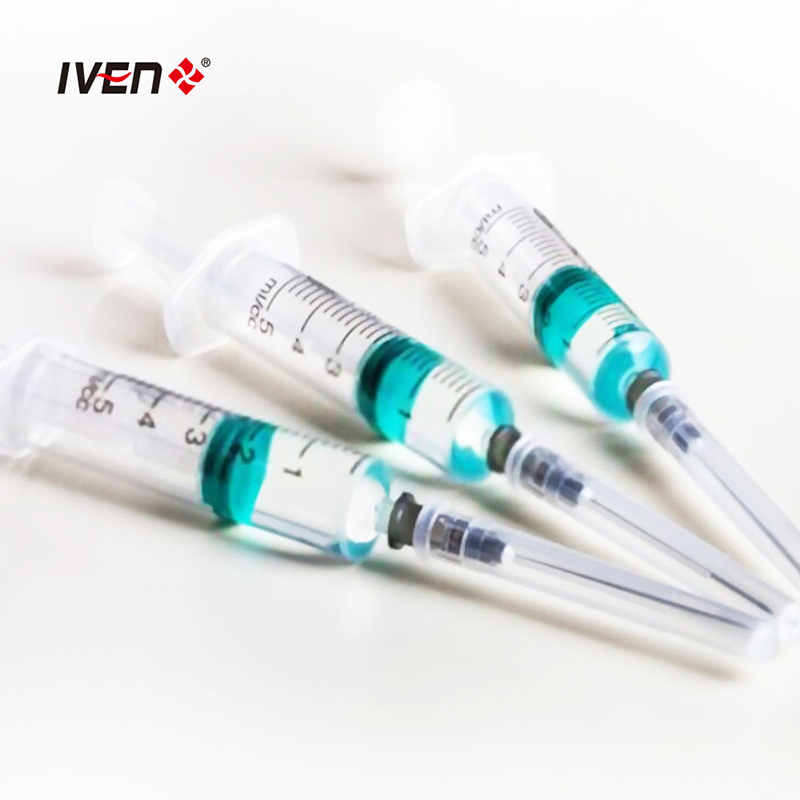
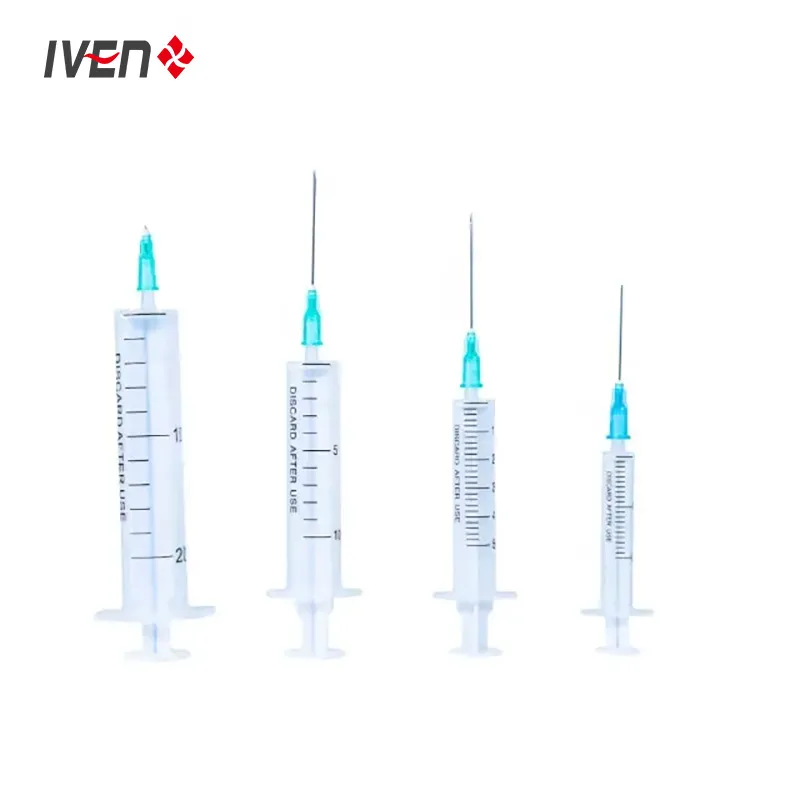
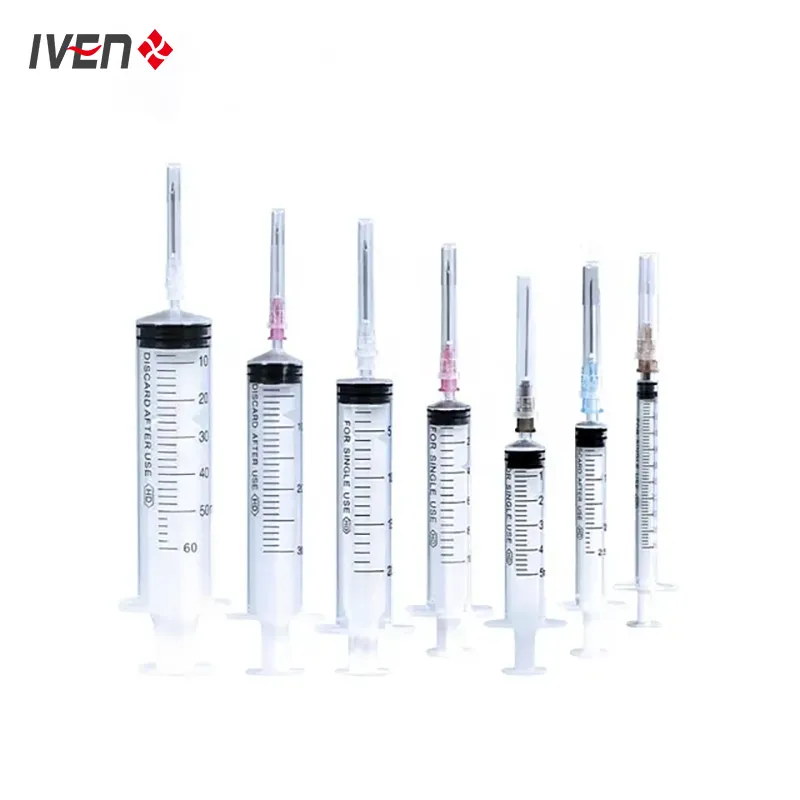
Disposable syringes are manufactured from high molecular weight polypropylene materials and have good biocompatibility. They are divided into three-piece and two-piece structures. The three-piece structure consists of a core bar, a rubber stopper, outer sleeves and an injection needle and outer packaging. The two-piece structure consists of a core bar, outer sleeves, injection needle and outer packaging.
Medical needles and syringes should be sterilised with ethylene oxide. Sterile, no heat source. Qualified products should have high transparency, clear and accurate graduation marks, and sliding performance should be good. The end of the tube structure should be designed with an anti-drop structure to prevent the mandrel from falling out of the tube; the core rod should be designed with a self-destructive structure to facilitate harmless treatment after single use.








Automatically stop for lack of material, automatically open for adding of material.
Consists of high-quality feeding system and assembly mechanism.
Feeds 4 components of syringe (plunger/ stopper/ needle/ barrel) to the assembly mechanism.
Completes assembly of plunger with rubber stopper, barrel with needle, and final combined assembly.
Greatly improve pass rate and production quality with machinery designed for endless improvement.
Response within 2 hours after receiving service requirements notification.




| Specification | 1ml | 2-3ml | 5ml | 10ml | 20ml | 30ml | 50ml |
|---|---|---|---|---|---|---|---|
| Production Speed | 180pcs/min | 200pcs/min | 250pcs/min | 250pcs/min | 160pcs/min | 150pcs/min | 120pcs/min |
| Dimension (mm) | 4200×3000×2200 | 4200×3000×2200 | 4200×3000×2200 | 4200×3000×2200 | 4200×3000×2200 | 4200×3000×2200 | 4200×3000×2200 |
| Power | AC220V/2.5KW | AC220V/2.5KW | AC220V/2.5KW | AC220V/2.5KW | AC220V/2.5KW | AC220V/2.5KW | AC220V/2.5KW |






 Lenavax Nutrition
Lenavax Nutrition